BIOELECTRIC MEDICINE • OCULAR SURFACE REPAIR
Programmable bioelectric therapy for corneal repair
Galvanis is developing OcuRegen™, a topical bioelectric therapy designed to restore endogenous wound-field signaling in impaired corneal healing.
Lead indication: persistent corneal epithelial defects (PCED).
MECHANISM-CONFIRMED
Quantifiable PD biomarker gates lead selection.
FAST, OBJECTIVE ENDPOINTS
PCED supports fast trials with clear endpoints.
LOCAL DELIVERY
Designed for minimal systemic exposure.
Persistent corneal epithelial defects are vision-threatening lesions that fail to close under standard supportive care. When closure stalls, patients can escalate through bandage lenses, serum-based therapies, amniotic membrane, and other interventions while risk compounds. Galvanis is targeting this setting first because the endpoint is objective, the treatment is local, and the path to translational signal is unusually efficient.
100,000 cases/year; a refractory subset escalates to procedures.
When closure fails, cases step through bandage lenses and autologous serum/PRP, then advanced interventions.
Medicare charges ~$95.6M (2020) on sutureless amniotic membrane grafts.
Non-healing corneal defects threaten vision and drive procedural escalation
No topical therapy is approved specifically for persistent corneal epithelial defects in the United States.
Why Galvanis wins
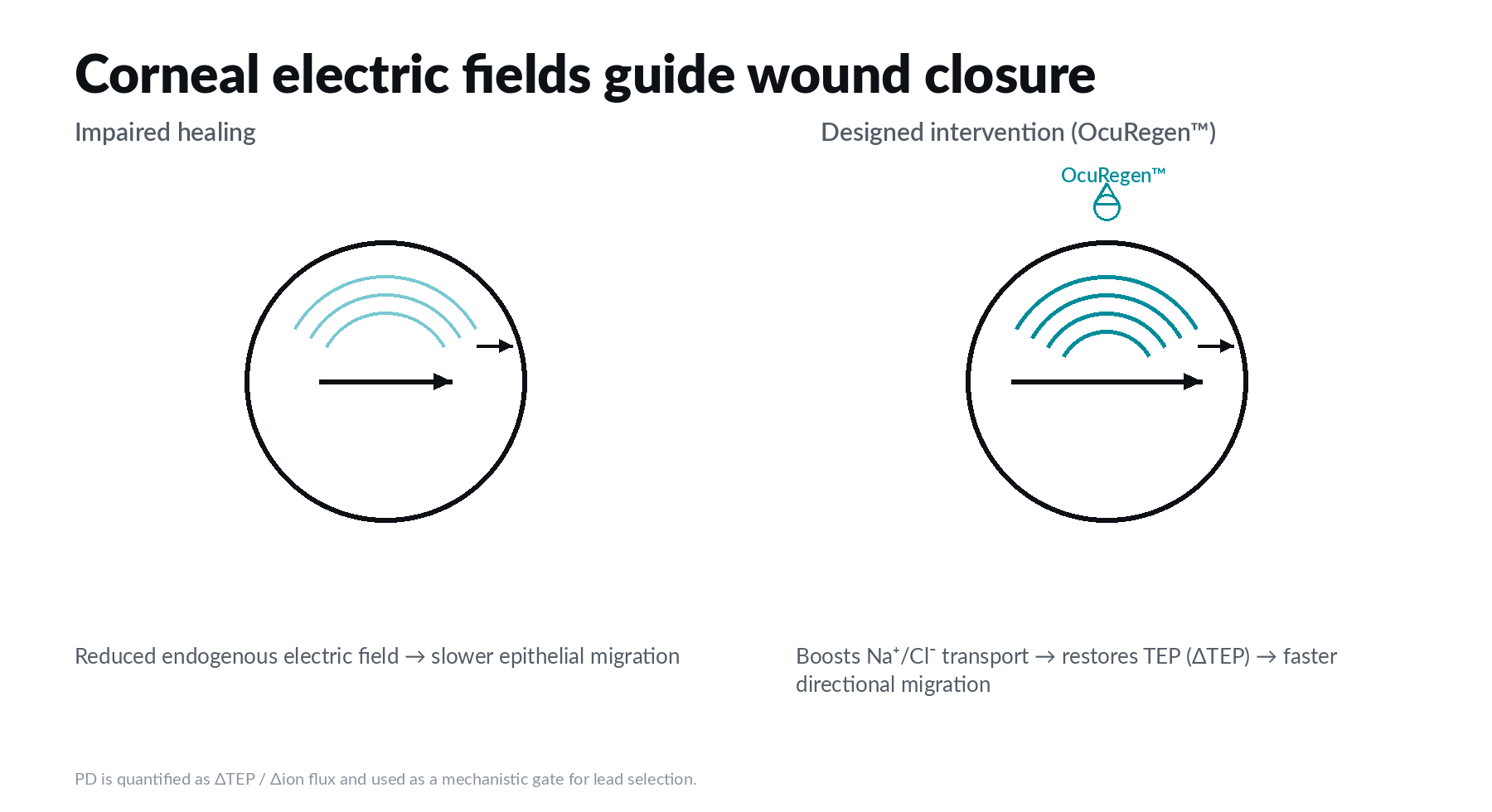
Corneal repair is not only biochemical. It is bioelectric.
The corneal epithelium maintains a transepithelial potential. When the tissue is wounded, endogenous electric fields form around the defect and help guide directional epithelial migration. Human donor corneas show these wound currents can be modulated pharmacologically. Galvanis is translating that control layer into a topical therapeutic program and advancing candidates only when they clear predefined pharmacodynamic and functional gates.
Pharmacodynamic-gated development with fast go/no-go decisions
We de-risk early by using a quantifiable biomarker (TEP restoration) to screen candidates quickly and avoid late-stage failures associated with variable or ‘black box’ modalities.
Investor snapshot
Key diligence items; full materials available upon request.
RAISE
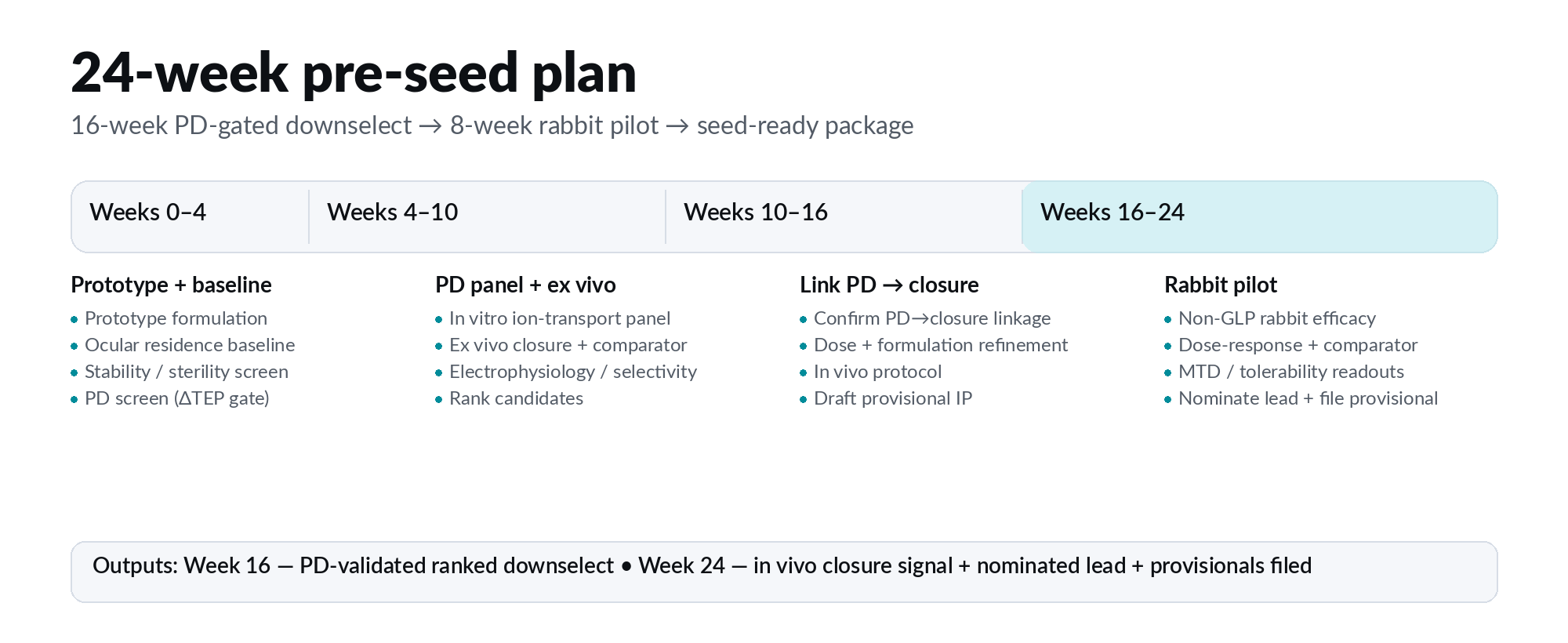
Galvanis is raising a $900K to run a 24-week, milestone-gated program designed to answer one question with speed and precision: can a topical bioelectric modulator produce a measurable pharmacodynamic signal and an in vivo closure signal in corneal repair?
MILESTONES
Week 16: PD-ranked lead downselect
Week 24: rabbit efficacy signal and lead nomination
ROUND OUTPUTS
Nominated lead
Rabbit efficacy package
Provisional IP filings
Seed-ready diligence package
Indications strategy
Team
Cody Rasmussen-Ivey, PhD
Founder & CEO
Bioelectricity & regenerative medicine scientist
Developed a DARPA ‘smart bandage’
Former Colossal
EXECUTION TEAM (FRACTIONAL, PRE-SEED)
CANDIDATE MANUFACTURING
Genscript; alternates Bachem and PolyPeptide
DRUG SCREEN
Charles River Labs; alternates Labcorp, Metrion Biosciences, and WuXi
IN VIVO OCULAR MODEL
Labcorp; alternates WuXi and Iris Pharma
Partner, invest, or collaborate
If you’re an investor, CRO, media, or scientist/medical professional, we’d like to talk.